medical data and annotation service for ai projects
Your Best Dataset is here
-
Organ
- Abdominal aorta
- Biliary system
- Bladder
- Bones
- Brain
- Bronchi
- Cervical Spine
- Colon
- Cystic artery
- Cystic duct
- Duodendum
- Esophagus
- Fallopian tubes
- Fatty tissue
- Femoral head
- Gallbladder
- Heart
- Heart and major vessels
- Kidney
- Liver
- Lumbar spine
- Lung
- Mediastinum
- Mesenteric artery
- Mesenteric vein
- Nasal sinus
- Ovary
- Pancreas
- Pelvic bones
- Prostate
- Pulmonary lobar vasculature
- Pulmonary lobes
- Pulmonary vasculature
- Rectum
- Rectum and Colon
- Rib
- Rib cage
- Sacrum
- Sigmoid colon
- Spine
- Spleen
- Stomach
- Thoracic aorta
- Thoracic veins
- Tracheobronchial tree
- Urethra
- Urinary bladder
- Uterus
- Vertebrae
- Vessels
- Organ System
-
Pathology
- Angiomyolipoma kidney
- Appendicitis
- Arthritis
- Brain tumor
- Bronchial tumor
- Cirrhosis
- Colon Cancer
- Colon diverticulitis
- Colon tumor
- Cystic structures
- Fibroma kidney
- Ischemic heart disease
- Kidney abscess
- Kidney cyst
- Kidney tumor
- Lipoma kidney
- Liver tumor and metastases
- Lung cancer
- Lung tumor
- Multicystic kidney dysplasia
- Oncocytoma kidney
- Osteoarthritis
- Osteochondrosis
- Pancreatic cancer
- Pancreatic cancer and benign lesions
- Pancreatitis
- Pelvic tumor
- Pneumonia
- Prostate Cancer
- Pulmonary embolism
- Pulmonary infection
- Rectal cancer
- Sigmoid colon tumor
- Uterine fibroids
- Test
- Video
-
 CT lungs with trachea, bronchi, and vessels dataset with annotation for ML AI
Get the CT lungs with trachea, bronchi, and vessels dataset with annotation for ML AI from Medical Data Cloud to improve algorithms of ML dataAge group: 18 and olderGender: All inclusiveData type: CTFormat: DICOM, jsonOrgan: Lungs, Trachea, Bronchi, Vessels
CT lungs with trachea, bronchi, and vessels dataset with annotation for ML AI
Get the CT lungs with trachea, bronchi, and vessels dataset with annotation for ML AI from Medical Data Cloud to improve algorithms of ML dataAge group: 18 and olderGender: All inclusiveData type: CTFormat: DICOM, jsonOrgan: Lungs, Trachea, Bronchi, Vessels -
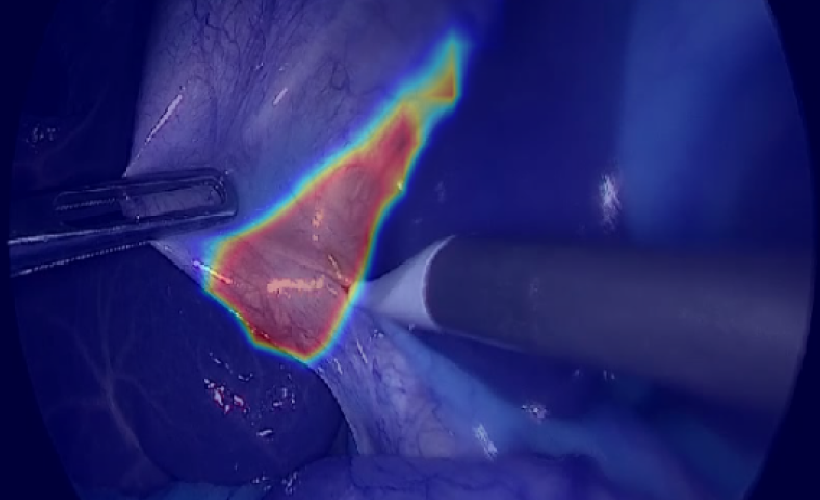
 Laparoscopic cholecystectomy dataset
Use laparoscopic cholecystectomy dataset from Medical Data Cloud to improve algorithms of ML data analysis and recognition of the gallbladder, liver, cystic, common hepatic ducts and other anatomical structures of the abdominal cavity.Age group: 18 and olderGender: All inclusiveData type: VideoFormat: mp4 H265 CodecOrgan: Gallbladder, liver, cystic, common hepatic ducts
Laparoscopic cholecystectomy dataset
Use laparoscopic cholecystectomy dataset from Medical Data Cloud to improve algorithms of ML data analysis and recognition of the gallbladder, liver, cystic, common hepatic ducts and other anatomical structures of the abdominal cavity.Age group: 18 and olderGender: All inclusiveData type: VideoFormat: mp4 H265 CodecOrgan: Gallbladder, liver, cystic, common hepatic ducts -
 Abdominal CT dataset with annotations for ML AI
Use abdominal CT dataset with annotations for ML AI from Medical Data Cloud to improve algorithms of ML data analysisAge group: 18 and olderGender: All inclusiveData type: CTFormat: DICOM, jsonOrgan: Abdomen
Abdominal CT dataset with annotations for ML AI
Use abdominal CT dataset with annotations for ML AI from Medical Data Cloud to improve algorithms of ML data analysisAge group: 18 and olderGender: All inclusiveData type: CTFormat: DICOM, jsonOrgan: Abdomen -
 Prostate MRI dataset
Obtain our vast prostate MRI dataset with clear organ images and accompanied detailed radiology reports, with and without pathology.Age group: 18 and olderGender: MaleData type: MRIFormat: DICOM, jsonOrgan: Prostate
Prostate MRI dataset
Obtain our vast prostate MRI dataset with clear organ images and accompanied detailed radiology reports, with and without pathology.Age group: 18 and olderGender: MaleData type: MRIFormat: DICOM, jsonOrgan: Prostate -
 MRI brain dataset with annotation for ML AI
Use MRI brain dataset with annotation for ML AI from Medical Data Cloud to improve algorithms of ML data analysisAge group: 18 and olderGender: All inclusiveData type: MRIFormat: DICOM, jsonOrgan: Brain
MRI brain dataset with annotation for ML AI
Use MRI brain dataset with annotation for ML AI from Medical Data Cloud to improve algorithms of ML data analysisAge group: 18 and olderGender: All inclusiveData type: MRIFormat: DICOM, jsonOrgan: Brain -
 Laparoscopic colectomy dataset
Get the laparoscopic colectomy dataset from Medical Data Cloud to improve algorithms of ML dataAge group: 18 and olderGender: All inclusiveData type: VideoFormat: mp4 H265 CodecOrgan: Colon
Laparoscopic colectomy dataset
Get the laparoscopic colectomy dataset from Medical Data Cloud to improve algorithms of ML dataAge group: 18 and olderGender: All inclusiveData type: VideoFormat: mp4 H265 CodecOrgan: Colon -
 ECG, myocardial ischemia and infarction dataset
Use the ECG, myocardial ischemia and infarction dataset from Medical Data Cloud to explore and run machine learning dataAge group: 18 and olderGender: All inclusiveData type: ECGFormat: jsonOrgan: Heart
ECG, myocardial ischemia and infarction dataset
Use the ECG, myocardial ischemia and infarction dataset from Medical Data Cloud to explore and run machine learning dataAge group: 18 and olderGender: All inclusiveData type: ECGFormat: jsonOrgan: Heart -
 Laparoscopic sleeve gastrectomy for morbid obesity dataset
Get the laparoscopic sleeve gastrectomy for morbid obesity dataset from Medical Data Cloud to improve algorithms of ML data analysis and recognition of the liver, spleen, stomach and other anatomical structures of the abdominal cavity.Age group: 18 and olderGender: All inclusiveData type: VideoFormat: mp4 H265 CodecOrgan: Stomach, esophagus
Laparoscopic sleeve gastrectomy for morbid obesity dataset
Get the laparoscopic sleeve gastrectomy for morbid obesity dataset from Medical Data Cloud to improve algorithms of ML data analysis and recognition of the liver, spleen, stomach and other anatomical structures of the abdominal cavity.Age group: 18 and olderGender: All inclusiveData type: VideoFormat: mp4 H265 CodecOrgan: Stomach, esophagus
Medical Data Cloud is a leading Medical Data Marketplace for AI projects, which provides large, medium and small enterprises with an unparalleled access to extensive amount and variety of Medical Data.
Our customers In Radiology, Cardiology, Robotic Surgery, Telemedicine, and other healthcare areas need and use large amount of quality imaging and other medical data, which we deliver.
Medical Data Cloud actively invests in the on-going data security and privacy. The company works with the best on the market cybersecurity firms and data protection tools.
We understand that success of our customer’s ML/AI projects depends on the availability of right datasets. We, therefore, work tirelessly to deliver the right datasets for your ML/AI needs on time and budget to become your reliable data partner.